If you’ve heard of hepatitis, you probably know it has something to do with the liver. But there’s a whole family of hepatitis viruses, each with its own personality when it comes to how it spreads, what it does to your body, and how we can prevent or treat it. Let’s walk through the three most common types—hepatitis A, B, and C—and then dive into a controversy that’s making headlines right now: the hepatitis B vaccine.
What Is Hepatitis, Anyway?
At its core, hepatitis just means inflammation of the liver. Your liver is a workhorse organ that filters toxins, produces essential proteins like albumin, processes amino acids, and stores energy. When a hepatitis virus attacks it, the inflammation can range from a minor inconvenience to a life-threatening condition. The three main culprits—hepatitis A, B, and C viruses—are completely different organisms that just happen to target the same organ.
Hepatitis A: The Food and Water Troublemaker
Hepatitis A is often called “traveler’s hepatitis” because it spreads through food and water that are contaminated with fecal matter. Think of it as the virus you might pick up from eating unwashed produce, drinking contaminated water, or consuming raw shellfish from polluted waters. Other risk factors include unprotected sex and IV drug use. According to the CDC, there were an estimated 3,300 acute infections in 2023 in the United States.
The good news about hepatitis A is that it typically heals itself within 2 months. When symptoms appear—which take about 15 to 50 days after infection—they can include jaundice (that yellowing of the skin and eyes), fever, fatigue, nausea, and dark urine. Many young children don’t show any symptoms at all. The virus doesn’t become chronic, and once you’ve had it, your body produces antibodies that protect you for life.
Prevention is straightforward: there’s a safe and effective vaccine, and basic hygiene goes a long way. Wash your hands thoroughly, especially after using the bathroom and before preparing food. When traveling to areas with questionable water quality, stick to bottled or boiled water and avoid washing raw food in local water.
Treatment is mostly supportive—rest, fluids, and time. Your liver does the healing work itself.
Hepatitis B: The Blood and Body Fluid Virus
Hepatitis B is where things get more serious. This virus spreads through blood and other body fluids, which means it can be transmitted through sexual contact, sharing needles, or from mother to baby during childbirth. Healthcare workers are especially at risk from needle sticks and sharps injuries. It’s a highly infectious and tough virus that can live on surfaces for up to a week. Even tiny amounts of dried blood on seemingly innocent things like razors, nail clippers, or toothbrushes can potentially spread the infection.
According to the CDC, there were an estimated 14,400 acute infections in 2023, Approximately 640,000 adults were living with chronic hepatitis B during the 2017-2020 period and that’s what makes it particularly concerning: while the hepatitis B virus often causes short-term illness, it can become chronic.
The incubation period is long—typically 90 days with a range of 60 to 150 days. When symptoms do appear, they mirror hepatitis A: jaundice, fatigue, abdominal pain, nausea, and dark urine. But here’s the frightening part: most young children and many adults show no symptoms at all, meaning they can spread the virus without knowing they’re infected.
The chronic infection risk varies dramatically by age. If you’re infected as a newborn, you have a 90% chance of developing chronic hepatitis B. For adults, the risk drops to under 5%. Those with chronic infection face serious long-term consequences—15% to 25% of people with chronic infection develop serious liver disease, including cirrhosis, liver failure, or liver cancer.
Treatment for acute hepatitis B is supportive, but several antiviral medications are available for people with chronic infection. These don’t completely eradicate the disease but produce a “functional cure” that significantly slows liver damage and reduces complications.
Prevention is critical. There’s a highly effective vaccine—we’ll talk more about the controversy surrounding it in a moment. Avoiding exposure to infected blood and body fluids is essential. This means safe sex practices, never sharing needles or personal care items that might have blood on them, and ensuring proper sterilization of medical and tattooing equipment.
Hepatitis C: The Silent Epidemic
Hepatitis C is transmitted primarily through blood-to-blood contact. The most common route is sharing needles among people who inject drugs, though it can also spread through contaminated medical equipment, and rarely through sexual contact. Mother-to-child transmission during childbirth is possible but uncommon. Screening of blood products has made transfusion related infections rare. About 10% of cases have no identified source.
What makes hepatitis C insidious is its stealthy nature. Many people with hepatitis C don’t have symptoms, and acute hepatitis with jaundice is rare, occurring in only about 10% of infections. The symptoms that do appear—fatigue, mild flu-like feelings—are easily dismissed. Meanwhile, the majority of people (60-70%) develop chronic infection. I recommend a screening blood test at least once for all adults over age 55, as they are the group most likely to have hepatitis C without an identifiable source.
The incubation period ranges widely, from 2 weeks to 6 months, typically 6 to 9 weeks. Without treatment, chronic hepatitis C can lead to cirrhosis and liver cancer over decades. Before modern treatments, it was a leading cause of liver transplants.
Treatment for hepatitis C has undergone a revolution. The old approach—interferon injections combined with ribavirin—had terrible side effects and worked in only about half of patients. Today, we have direct-acting antivirals (DAAs), which can cure more than 95% of cases with just 8-12 weeks of well-tolerated oral medication. These drugs target specific proteins the virus needs to replicate, essentially starving it out of existence. The treatment is so effective that hepatitis C is now considered a curable disease.
Prevention focuses on avoiding blood-to-blood contact. Never share needles, syringes, or any drug equipment. If you’re getting a tattoo or piercing, ensure the facility follows proper sterilization procedures. Healthcare workers should follow standard precautions with blood and body fluids. Unfortunately, there’s no vaccine for hepatitis C yet, though researchers continue working on one.
The Hepatitis B Vaccine Controversy: What’s Really Happening
Now let’s address the elephant in the room—the recent controversy over the hepatitis B vaccine for newborns. This topic exploded in the news in December 2025, and it’s worth understanding what’s currently going on versus what the science says.
The Recent Development
On December 5, 2025, the CDC’s Advisory Committee on Immunization Practices (ACIP) voted 8-3 to recommend hepatitis B vaccination at birth only for infants born to mothers who test positive for the virus or whose status is unknown. This reverses decades of policy that recommended universal hepatitis B vaccination for all newborns within 24 hours of birth.
The Arguments For Changing the Policy
Some ACIP members raised concerns about vaccine safety and parental hesitancy. Committee member Retsef Levi heralded the move as “a fundamental change in the approach to this vaccine,” which would encourage parents to “carefully think about whether they want to take the risk of giving another vaccine to their child”. The controversy includes historical concerns about possible links between the hepatitis B vaccine and conditions like multiple sclerosis, autism, and other autoimmune disorders.
What Science Actually Shows
The evidence on vaccine safety is quite robust. Concerns about multiple sclerosis emerged in France in the 1990s. Since then, a large body of scientific evidence shows that hepatitis B vaccination does not cause or worsen MS. The World Health Organization’s Global Advisory Committee on Vaccine Safety has concluded there is no association between the hepatitis B vaccine and MS. It is one of the safest vaccines studied.
As for other safety concerns, CDC reviewed VAERS reports from 2005-2015 and found no new or unexpected safety concerns. The most common side effects are minor: soreness at the injection site, headache, and fatigue lasting 1-2 days.
Why the Universal Birth Dose Matters
The scientific and medical communities have strongly opposed this policy change. The American Academy of Pediatrics states that from 2011-2019, rates of reported acute hepatitis B remained low among children and adolescents, likely explained in part by the implementation of childhood hepatitis B vaccine recommendations published in 1991.
Here’s why newborns are so vulnerable: infected infants have a 90% chance of developing chronic hepatitis B, and a quarter of those will die prematurely from liver disease when they become adults.
The “just target high-risk babies” approach has a major flaw: the CDC estimates about 640,000 adults have chronic hepatitis B, but about half don’t know they’re infected. Before universal vaccination, about half of infected children under 10 got it from their mothers—the rest contracted it through other exposures not identified by maternal screening.
The Global Context
Claims that the U.S. is an outlier don’t hold up. As of September 2025, 116 of 194 WHO member states recommend universal hepatitis B birth dose vaccination. European countries that do not recommend a universal birth dose have a much lower hepatitis B incidence rate and more robust antenatal maternal screening. The majority still recommend vaccination at two to three months.
The Bottom Line
All three types of hepatitis pose serious health risks, but we have powerful tools to prevent and treat them. Hepatitis A and B have safe, effective vaccines that have dramatically reduced disease rates. Hepatitis C, while lacking a vaccine, is now curable with modern antiviral medications.
The hepatitis B vaccine controversy highlights a broader tension in public health: balancing individual autonomy with community protection. The scientific evidence strongly supports the vaccine’s safety and the effectiveness of universal newborn vaccination in preventing a disease that can be fatal. Multiple studies, decades of safety data, and recommendations from medical organizations worldwide back this up.
For parents making decisions about their newborns, the facts are these: hepatitis B is a serious disease with a high risk of becoming chronic in infants, the vaccine is highly effective at preventing infection, and extensive safety monitoring has found it to be safe with only minor, temporary side effects. As hepatitis research continues, we’re seeing remarkable progress—from the near-eradication of hepatitis A in vaccinated populations to the transformation of hepatitis C from a chronic, often fatal disease to a curable one. These advances remind us how far we’ve come in understanding and combating these liver viruses.
Sources


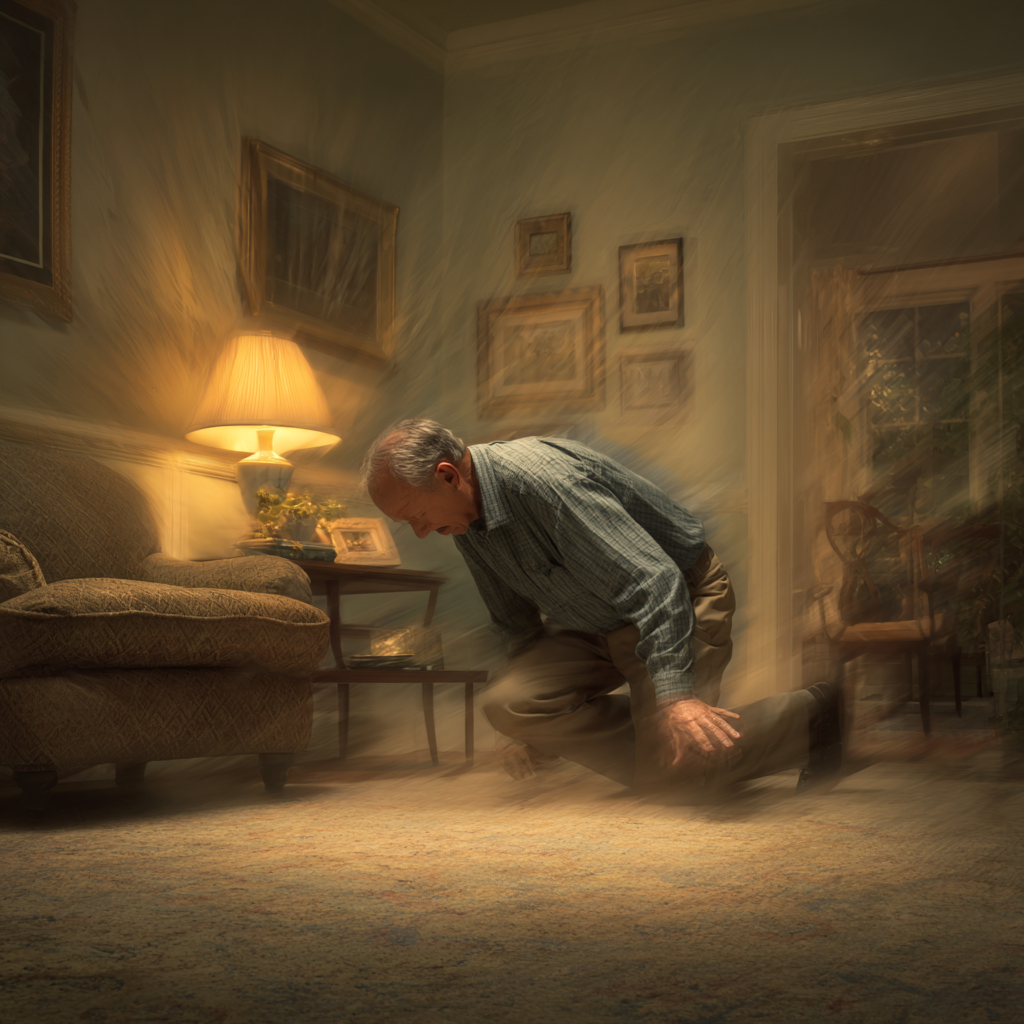
Understanding Medical Care Guidelines
By John Turley
On August 7, 2024
In Commentary, Medicine
An important discussion to have with your physician.
Trivia question: What are medical guidelines? Are they rules we must follow or are they simply suggestions or are they something in between?
As we get older and have more frequent visits to the doctor, we are bound to hear one of them say, “according to the guidelines”. To understand how the guidelines apply to you, it is important to know how and why they are developed. You also need to know if there are ever times when you shouldn’t follow them.
At the end of this article, I’ll tell you about my experience with one specific guideline, and how strictly following it possibly could have led to a bad outcome for me. But first, let’s learn a little more about medical guidelines.
Medical care guidelines, also called clinical guidelines, come in two general classes. There are guidelines for preventative care and guidelines for the management of disease processes.
Guidelines have several goals. They are intended to improve public health by recommending evidence-based preventive and treatment measures to help reduce the incidence and severity of disease and improve overall public wellbeing. They’re designed to optimize resource utilization by preventing unnecessary treatment and screening tests. They are also intended to reduce health care disparities by ensuring that all recommended treatments are widely available and are based on the most up-to-date evidence so that health care across the nation is at a uniformly high level of quality.
Sources of Guidelines
Preventative care guidelines have to do with such things as cancer screening, cardiovascular health, vaccinations and immunizations, and lifestyle improvement such as diet and exercise recommendations. Disease management guidelines are developed to ensure the best possible treatment for diseases such as hypertension, diabetes and pulmonary disease.
Guidelines are developed by physician groups such as the American College of Physicians and the American Academy of Pediatrics. They are also developed by advocacy groups such as the American Cancer Society and the American Diabetes Association. Government organizations such as the Centers for Disease Control and Prevention and the National Institutes of Health also develop and promulgate medical care guidelines.
The United States Preventative Services Task Force (USPSTF) is an independent panel of experts in prevention and evidence-based medicine. They issue recommendations on a wide range of preventive services including screenings, counseling and preventative medications. The USPSTF rates medical care recommendations from Grade A, those with a high certainty of substantial benefits, all the way to Grade D, those services that are not recommended due to having no benefit or having harm that outweighs benefits. Their recommendations can be viewed at www.uspreventiveservicestaskforce.org.
Preventative care guidelines
Preventive care guidelines are designed to help identify and mitigate potential health issues before they become significant problems. They help to ensure adequate screening for significant disease processes. They are also designed to help avoid unnecessary screening which may lead to unnecessary treatment and cost.
Preventative care guidelines include such things as mammogram recommendations, colonoscopy recommendations, blood pressure and cholesterol screening, and prostate cancer screening. Preventative care guidelines also include recommendations for vaccinations both for children and adults. Recommendations on diet and the use of vitamins and supplements are one area where the guidelines seem to change frequently.
Treatment guidelines
Treatment guidelines provide a roadmap for managing specific medical conditions. These recommendations encompass diagnostic procedures, therapeutic interventions, and follow-up care to ensure optimal patient outcomes.
Treatment guidelines include recommendations for such things as initiation of blood pressure management and diabetes management. They provide recommendations for diagnostic modalities and specific medications and dosages.
For example, treatment guidelines include blood pressure levels at which medication should be started, the goal of treatment and specific medication, depending on what other medical conditions the patient may have. Similarly, there are blood glucose management recommendations for diabetics that are tailored to specific patient populations. The use of bronchodilators and pulmonary rehabilitation and oxygen therapy for lung diseases are also the subject of a series of guidelines. Treatment guidelines continually evolve as new medications are developed and our understanding of disease processes improves.
Understanding the variability in guidelines.
While the guidelines developed by the various organizations share a common goal of improving patient care, their methodologies and focus areas can differ, reflecting diverse perspectives and priorities within the medical community. There’s not a single set of guidelines that are fixed across all specialties. While the various guidelines are generally in agreement, some may have slightly different recommendations for such things as the onset and aggressiveness in treating hypertension or diabetes. There may be variations in the guidelines for diagnostic testing such as mammograms or colonoscopies. For example, the USPSTF recommends biennial mammograms for women aged 50 to 74, whereas the American College of Surgeons advises annual mammograms starting at age 45 and transitioning to biennial screening at 55. The discrepancy lies in differing interpretations of the balance between benefits and harms of more frequent screenings.
Some guidelines may also become outdated, not reflecting new medications or new treatment plans. Even where there are variations, all guidelines strive to be evidence based, patient centered, and up to date.
Additionally, guidelines need to be individualized to meet the needs of each patient. The overall guidelines are based on the most effective health care for the population as a whole. Some patients may require specialized screening or treatment. For example, women who have a family history of early onset of breast cancer or of genetic mutations may require screening at an earlier age or more frequent screening. Men with a family history of prostate cancer at a young age or of a particularly aggressive prostate cancer may require earlier screening including biopsies or may need screening beyond the age that general guidelines recommend screening is no longer necessary.
My Experience
Several years ago, I received a diagnosis no one wants to hear. Cancer! Prostate cancer to be specific. Thanks to two skilled urologists, I’ve been cancer free for five years.
But it might not have had a happy ending. Please indulge me and let me tell you my story. I think it will be worth your time.
It starts with the PSA, the prostate specific antigen. This is something every man over 40 should know about and every man over 50 should consider getting checked.
So, what is the PSA? It is a protein that is produced by both cancerous and normal cells of the prostate gland. It can be elevated by prostate cancer but it can also be elevated by prostatitis (an infection of the prostate) or an enlarged prostate (benign prostatic hypertrophy). It is checked through a simple blood test your family doctor can order as part of your annual work up.
What are the recommendations for the PSA? The USPSTF has the following three recommendations: (1) consideration of annual screening for men aged 55 to 69 with no family history of prostate cancer; this should be a shared, informed decision between the patient and his physician; (2) for men who have a significant family history of prostate cancer consideration should be given to screening beginning at age 40; (3) for men over 70 years old they recommend against screening for prostate cancer. Please note the phrase “consideration of screening”. This is not a firm recommendation.
A PSA test can have false positives that may lead to unnecessary biopsies or surgery. Only about 25% of men who have a prostate biopsy are found to have cancer. Although, it is important to recognize that a prostate biopsy does not test the entire gland. It takes samples from several areas of the prostate. It is possible, though unusual, that a cancer could be missed in the biopsy process
Additionally, most prostate cancer is very slow growing. Most men who have prostate cancer later in life will generally die of something else before they would die of prostate cancer. However, a small percentage of men will have a high-grade prostate cancer that can progress rapidly and cause their death.
I’m going to use my personal experience as a way of explaining why it is important to have a discussion with your physician about guidelines. The week before my 70th birthday I went in to get my annual physical. In our clinic we have a “birthday panel”, a set of blood tests that we draw for people annually for their physical exam. I had not planned to have my PSA checked since it was not recommended by either the USPSTF or the American Academy of Family Physicians for 70-year-olds. However, it had slipped my mind that a PSA was part of our “birthday panel”.
My PSA came back slightly elevated. Since it was a very minor elevation, I followed the guidelines and waited six months and repeated it. At that time, it increased only a small amount. The guidelines suggested repeating it again in six months. I have to admit though, I have never been a wait-and-see kind of guy. I scheduled an appointment with a urologist.
The urologist and I discussed the options. He told me that the elevation was slight, and we could wait and repeat it in 6 months or if I wished we could do a biopsy. I decided on a biopsy and then after receiving the biopsy results and having further discussions, I eventually decided on surgery. It was my decision, as it should be, made in consultation with my physician and my family.
The post-operative pathology report said that there was a high-grade carcinoma that apparently had been missed by the biopsy. It had extended beyond the capsule of the prostate. Fortunately for me it had not metastasized and had not spread to the lymph nodes, nor had it extended beyond the fat layer surrounding the prostate. Had I followed the guidelines and waited another year or even six months for a repeat biopsy, it is possible that the outcome may have been different.
What’s the bottom line?
Does my experience mean that the guidelines should be ignored? Far from it, I made an informed decision, in conjunction with my physician, on what was best for me. Additionally, I have followed the guidelines in the management of my hypertension and high cholesterol.
Healthcare guidelines are essential in promoting preventive care and effective treatment and in helping clinicians provide high-quality, evidence-based care. But the guidelines are just that, guidelines they are not “set in stone” rules for healthcare. It’s important for you to discuss your health care with your physician. Be an informed health care consumer. Ask how the guidelines are being used to manage your health care and how they may be affected by your family history or personal history. You and your physician should be involved in joint decision making. Your individual plan will generally follow the guidelines while having some variation based on what is the best care for you. And that’s what the guidelines are all about, making sure we are able to provide the best possible health care for all of our citizens.